|
11/6/2022 0 Comments Clo2 molecular geometry 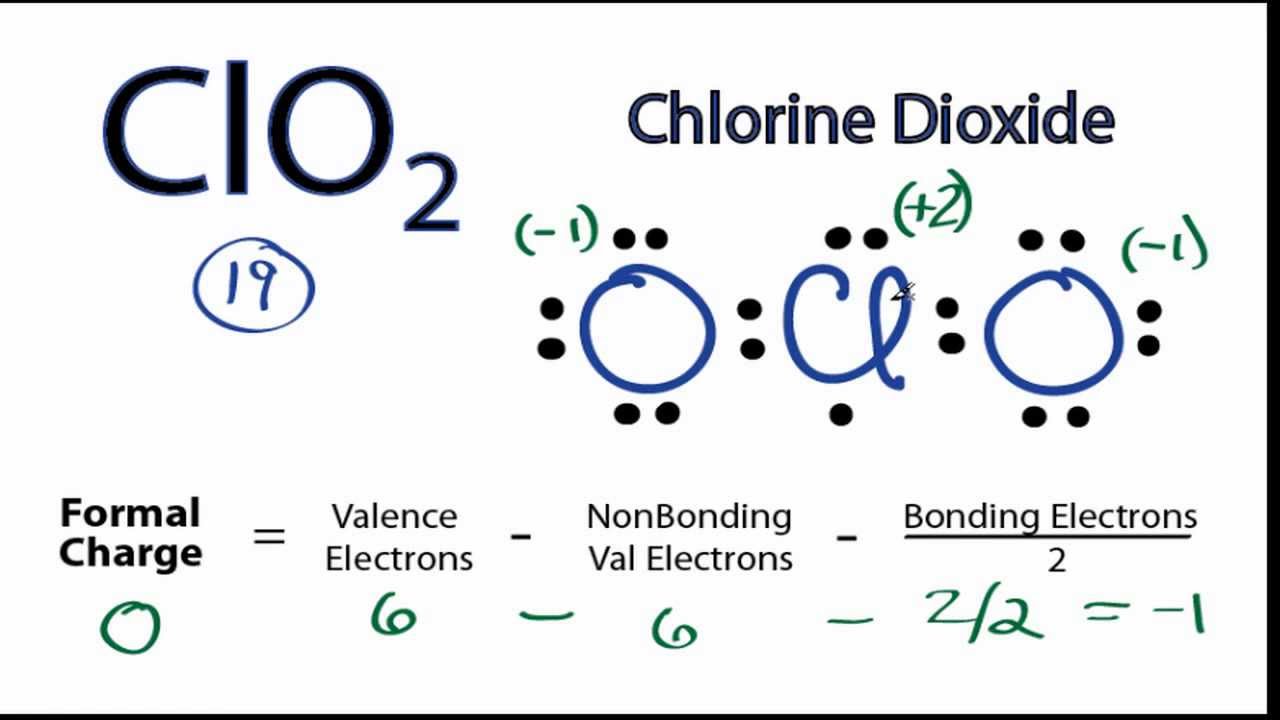 This can be studied in the detail with the help of the Valence Shell Electron Pair Repulsion (VSEPR) Theory or the AXN method.Īccording to this theory, the presence of two lone pairs of valence electrons on the chlorine atom exerts force and bends the structure giving the bond angle slightly lesser than 109°. It is bent because of the lone pairs of valence electrons on the chlorine atom and the uneven bond type between each chlorine and oxygen atom. Moreover, the atom with the lowest electronegativity value becomes the central atom as it has to make the maximum number of bonds.Īs a result, chlorine becomes the central atom.įrom the Lewis structure, it is clear that chlorine dioxide or chlorite ion is a triatomic molecule that is bent in shape. The explanation for the same lies in the formal charge distribution that compels the formation of a double bond and a single bond.Įven though the new structure is stable, it is the oxygen atom that usually undergoes bond formation with another nearby atom.Ĭhlorine is the central atom because it is present as the single entity within the chlorine dioxide molecule. One single bond is formed between chlorine and oxygen atoms and a double bond is formed between chlorine and another oxygen atom. 
Step 4: Find the type of bond formation taking place in one chlorine oxide. Studying the formal charge distribution in detail also gives us the reason behind the double bond forming between one oxygen and chlorine atom. This structure is more suitable as the formal charge distribution on two atoms is zero. Let us find the formal charge distribution of the above-mentioned molecule. Let us check the correct lewis structure of the ClO2- molecule as below. Therefore the above structure is unstable because there exists another Lewis structure where the formal distribution of some participating atoms is zero. With this, -1 formal charge still remains on another oxygen atom. Here +1 formal charge of the chlorine atom cancels out the -1 formal charge of one of the oxygen atoms. Now we need to calculate the formal charge distribution on chlorine dioxide molecule:įormal Charge = Valence Electrons – Non-Bonding Electrons – ½ Bonding Electrons The reason for the same is each molecule wants to achieve a stable structure that can be gained only with zero formal charge distribution. 
The aforementioned diagram is one of the lewis structures of chlorine dioxide but, we need to check its stability with the help of formal charge distribution. Step 3: Draw the skeleton of chlorine dioxide. Hence, with no anomaly, the total valence electrons required by one chlorine dioxide would have been 24 which is now 26 with the anomaly of expanded octet in chlorine atom. The third group has the ability to expand its octet and accommodate more valence electrons to achieve a stable molecular structure. CLO2 MOLECULAR GEOMETRY FULLIt is a classic example of chemistry being full of exceptions!Ĭhlorine belongs to the third group in the periodic table. Here, an anomaly exists with the chlorine atom as it can increase its octet to accommodate more valence electrons. Step 2: Find the valence electrons further needed to stabilize the molecule.īy the octet rule, each element needs eight valence electrons to complete its electronic configuration. So, the total number of valence electrons available in one chlorine dioxide or chlorite molecule is 20. There are two oxygen molecules in chlorine dioxide so the total is 19.īut chlorine dioxide exists as ClO2- during the formation of a bond, therefore we have one more valence electron available. It is 20 as chlorine has 7 valence electrons and oxygen has 6 valence electrons. Step 1: Find the total valence electrons in one molecule of chlorine dioxide. Let us follow some steps to draw the Lewis structure of chlorine dioxide: We will draw the Lewis structure of chlorine dioxide with 20 valence electrons. Due to this, the valence electrons in chlorine dioxide or chlorite are 20. This arises from the fact that chlorine dioxide is an unstable molecule and mainly exists as ClO2- during bond formation. 
Here, it is crucial to understand that chlorine dioxide is a strong anion and oxidizing agent. It might confuse many people as ClO2 comprises 19 valence electrons only. Now, it will be easier to draw the Lewis structure of chlorine dioxide as we know that there are 20 valence electrons in one chlorine dioxide molecule. To begin drawing the Lewis structure of Chlorine dioxide, first, it is essential to draw one for the participating elements.Įlectronic Configuration = 1s2 2s2 2p6 3s2 3p5 The Lewis structure is a pictorial representation of valence electrons taking part in the formation of bonds to produce a new molecule with new properties altogether. Conclusion Lewis Structure of Chlorine Dioxide (ClO2-) 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
