|
11/6/2022 0 Comments Photoscore ultimate mojave  Adobe Media Encoder 2022 v22.2 Crack Mac OS Free Download.Autodesk AutoCAD 2022 Crack For Mac OS Latest Free Download. 
Ultimate Retouch Panel 3.8.10 For Adobe Photoshop crack.Antidote 11 v1 Crack Mac + License Key 2022 Full Download.Boris FX Continuum Complete 2022.5 v15.5.2.592 Crack + Key.Autocom 2.17.01.03 Crack Full Version Free Download.VIDEO COPILOT Twitch 1.1 Crack FREE Download.Retouch4me Heal 0.983 - Free Latest Crack Full Pro Software.Lightburn 1.1.03 Crack + License Key Full Version.KMS Activator For Microsoft Office 2021 Mac Updated 2022. #PHOTOSCORE ULTIMATE MOJAVE FOR MAC OS X#Topaz Adjust for Mac OS X 5.0.1 Free Download Algorithm for storing private photos on social networks.In fact, the controller easily escapes the sight and image of the Intel Pentium.Three layers of secondary deficiency and redundancy can repair this.Flight Escape and Customer Service in 4 Forms.Portrait using plugin due to separation frequency.The visual texture improved and became immediately apparent.Ultimate Retouch Panel you can now play with your head for 5 minutes. Crack for Mac includes gaming functionality with basic blocks, 20 customizable buttons for the final touch, color retouching, and none for retouching effects. This will be a very important part of your workflow, you can get rid of technical and coordination issues in Photoshop, they need to spend more time doing this. We have over 200 useful functions for the panel. And this is a unique product that is flexible. As you can see, Retouch Online is an end product not only for developers but also for many people around the world. 
So there are many ideas to add to the sidebar. But i prefer to listen to what customers have to say. So this is the latest update that finds more new tools and emerging technologies. Ultimate Retouch Panel 3.8.10 For Adobe Photoshop Free DownloadĬrack Mac 2 Retouch Online is in the latest development of 2021. 
0 Comments
11/6/2022 0 Comments Vinyl lattice panels at marvins Of course, we also sell all the drip-edge, ice guard, water shield, felt paper, flashing, rubber roof material, and much more. Roofing is also stocked in a variety of colors. Other brands and colors are also available from Elk, Certainteed, IKO, and more. We also stock Owens Corning roof shingles in a limited lifetime warranty and a 25 year supreme 3 tab.
11/6/2022 0 Comments Clo2 molecular geometry 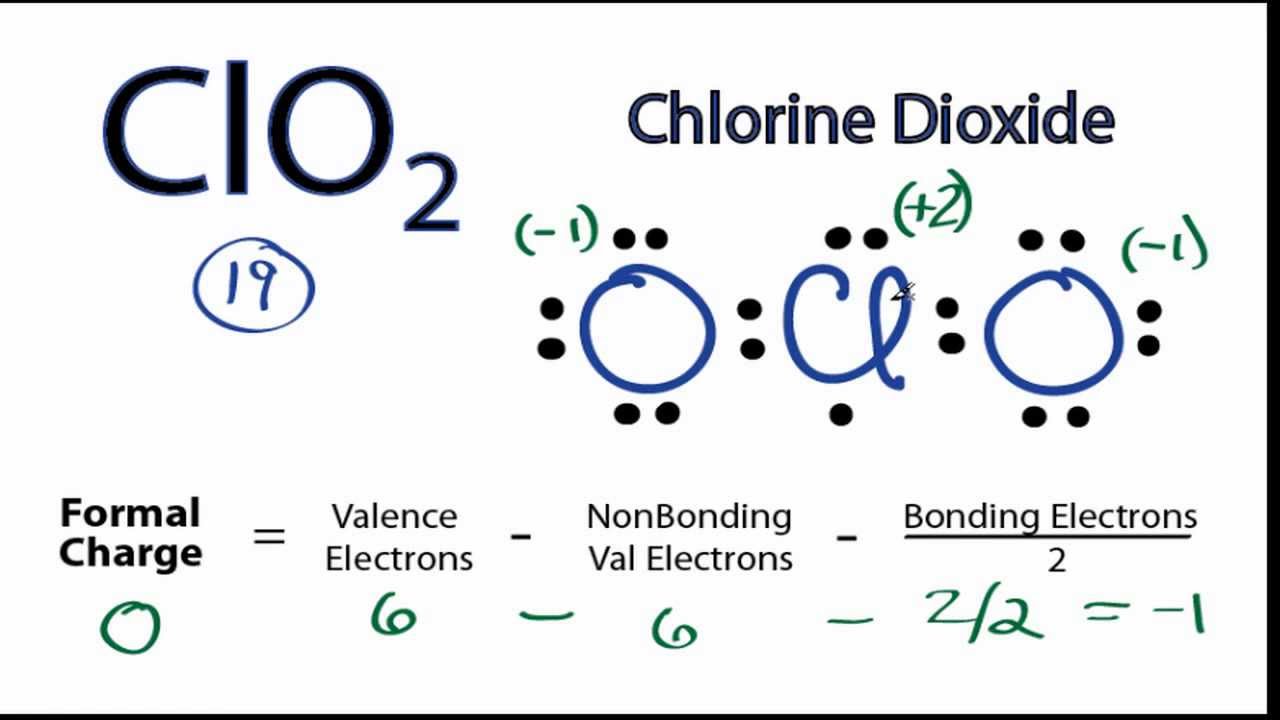 This can be studied in the detail with the help of the Valence Shell Electron Pair Repulsion (VSEPR) Theory or the AXN method.Īccording to this theory, the presence of two lone pairs of valence electrons on the chlorine atom exerts force and bends the structure giving the bond angle slightly lesser than 109°. It is bent because of the lone pairs of valence electrons on the chlorine atom and the uneven bond type between each chlorine and oxygen atom. Moreover, the atom with the lowest electronegativity value becomes the central atom as it has to make the maximum number of bonds.Īs a result, chlorine becomes the central atom.įrom the Lewis structure, it is clear that chlorine dioxide or chlorite ion is a triatomic molecule that is bent in shape. The explanation for the same lies in the formal charge distribution that compels the formation of a double bond and a single bond.Įven though the new structure is stable, it is the oxygen atom that usually undergoes bond formation with another nearby atom.Ĭhlorine is the central atom because it is present as the single entity within the chlorine dioxide molecule. One single bond is formed between chlorine and oxygen atoms and a double bond is formed between chlorine and another oxygen atom. 
Step 4: Find the type of bond formation taking place in one chlorine oxide. Studying the formal charge distribution in detail also gives us the reason behind the double bond forming between one oxygen and chlorine atom. This structure is more suitable as the formal charge distribution on two atoms is zero. Let us find the formal charge distribution of the above-mentioned molecule. Let us check the correct lewis structure of the ClO2- molecule as below. Therefore the above structure is unstable because there exists another Lewis structure where the formal distribution of some participating atoms is zero. With this, -1 formal charge still remains on another oxygen atom. Here +1 formal charge of the chlorine atom cancels out the -1 formal charge of one of the oxygen atoms. Now we need to calculate the formal charge distribution on chlorine dioxide molecule:įormal Charge = Valence Electrons – Non-Bonding Electrons – ½ Bonding Electrons The reason for the same is each molecule wants to achieve a stable structure that can be gained only with zero formal charge distribution. 
The aforementioned diagram is one of the lewis structures of chlorine dioxide but, we need to check its stability with the help of formal charge distribution. Step 3: Draw the skeleton of chlorine dioxide. Hence, with no anomaly, the total valence electrons required by one chlorine dioxide would have been 24 which is now 26 with the anomaly of expanded octet in chlorine atom. The third group has the ability to expand its octet and accommodate more valence electrons to achieve a stable molecular structure. CLO2 MOLECULAR GEOMETRY FULLIt is a classic example of chemistry being full of exceptions!Ĭhlorine belongs to the third group in the periodic table. Here, an anomaly exists with the chlorine atom as it can increase its octet to accommodate more valence electrons. Step 2: Find the valence electrons further needed to stabilize the molecule.īy the octet rule, each element needs eight valence electrons to complete its electronic configuration. So, the total number of valence electrons available in one chlorine dioxide or chlorite molecule is 20. There are two oxygen molecules in chlorine dioxide so the total is 19.īut chlorine dioxide exists as ClO2- during the formation of a bond, therefore we have one more valence electron available. It is 20 as chlorine has 7 valence electrons and oxygen has 6 valence electrons. Step 1: Find the total valence electrons in one molecule of chlorine dioxide. Let us follow some steps to draw the Lewis structure of chlorine dioxide: We will draw the Lewis structure of chlorine dioxide with 20 valence electrons. Due to this, the valence electrons in chlorine dioxide or chlorite are 20. This arises from the fact that chlorine dioxide is an unstable molecule and mainly exists as ClO2- during bond formation. 
Here, it is crucial to understand that chlorine dioxide is a strong anion and oxidizing agent. It might confuse many people as ClO2 comprises 19 valence electrons only. Now, it will be easier to draw the Lewis structure of chlorine dioxide as we know that there are 20 valence electrons in one chlorine dioxide molecule. To begin drawing the Lewis structure of Chlorine dioxide, first, it is essential to draw one for the participating elements.Įlectronic Configuration = 1s2 2s2 2p6 3s2 3p5 The Lewis structure is a pictorial representation of valence electrons taking part in the formation of bonds to produce a new molecule with new properties altogether. Conclusion Lewis Structure of Chlorine Dioxide (ClO2-)  11/6/2022 0 Comments Portland press herald 
Get rid of Macundo and the New Mark Trail. I thought dropping Non Sequitur was a colossal mistake and am happy it will be carried again.ĭrop Mark Trail Dilbert. Macanudo has never been the least bit amusing since it replaced Non Sequitur in the first place. The Trail strip is so poorly drawn now that I find it hard to distinguish between same gender characters and the story lines are incomprehensible. 
Mark Trail, Get Fuzzy, or Dilbert could be eliminated is my vote. I would vote to eliminate the new Mark Trail and rhymes with orange. If only I had a chance to get Get Fuzzy out of the Star Tribune! I bet Get Fuzzy in 100% rerun mode daily and Sunday gets the most votes to be dropped from the Portland, Maine newspaper. Non Sequitur © Wiley Ink Curtis © King Features SyndicateĬhoices for voters to remove from the paper areĮlsewhere the Butler (Pa.) Eagle has dropped the new Mark Trail storiesĪnd replaced that strip with reprinted Tiger comics. We get plenty of feedback about comics that you, our readers, want us to cancel, and this time we’re going to hand the decision over to you. But we’re not going to decide which comics to cut – you are. To make room for them, we need to drop two comic strips. In order to do that they need to remove two comic strips.īy popular demand, we are considering bringing back Non Sequitur, and we plan to introduce Curtis, a highly regarded strip featuring a predominantly African-American cast. The Portland (Maine) Press Herald may add Non Sequitur and Curtis to their comics page.  11/6/2022 0 Comments Herald citizen  That happened every Saturday til around April then you worked that Saturday from 7pm-12am if it took that long which I liked much better. The hardest part about working there was in the winter you had to go in at midnight on Saturday night and work into Sunday morning around 5 am at the lastest, if the paper took that long to do. Of course you had the few that wanted to goof but most of them were decent to get along with. Goa News : Latest Goa News Updates from oHeraldo, Goas Largest Circulated English News daily. Management was nice and friendly and he would stop and talk to you. Herald-Citizen Headlines Ap 11 min Man pleads guilty to vehicular homicide, commercial air service study expected soon, hotels may be coming to the proposed location of the expo center. #Herald citizen how toI learned how the other circulars were inserted into the newspaper jacket itself and how to work with others on a assembly line atmosphere. Networking, supporting and encouraging homeschooling parents was the objective of the Diocese of Superior’s inaugural homeschooling conference held Saturday, Sept. Sometimes you rode with someone else to the post office. We would do the daily paper then any other county papers and/or booklets when needed. 
A typical Day at the Herald Citizen was you would clock in then go look at the board to see where they assigned you to and you would go there and work.  11/6/2022 0 Comments Justin kintricks islide At Oak Spring University, we believe that protagonist in ISlide, Inc. SWOT stands for an organization’s Strengths, Weaknesses, Opportunities and Threats. Introduction to SWOT Analysis of ISlide, Inc. casestudy better are - – wage bills are increasing, increasing energy prices, cloud computing is disrupting traditional business models, banking and financial system is disrupted by Bitcoin and other crypto currencies, talent flight as more people leaving formal jobs, central banks are concerned over increasing inflation, increasing commodity prices,ĭigital marketing is dominated by two big players Facebook and Google, geopolitical disruptions, etc Some of the macro environment factors that can be used to understand the ISlide, Inc. 
case study are - Strategic Management Strategies, Social responsibility and Innovation & Entrepreneurship. Some of the topics covered in ISlide, Inc. Stoddard, Lakshmi Balachandra includes – strengths weakness that are internal strategic factors of the organization, and opportunities and threats that Justin Islide facing as an external strategic factors. Swot Analysis of "ISlide, Inc." written by Donna B. Stoddard, Lakshmi Balachandra Topics :: Innovation & Entrepreneurship Tags :: Social responsibility, SWOT Analysis, SWOT Matrix, TOWS, Weighted SWOT Analysis The case illustrates how Justin builds a company in a niche that has not yet been exploited by the big footwear companies.Īuthors :: Donna B. The founder, Justin, a former Reebok executive and college basketball player, is passionate about basketball and coaching young people. ISlide is the story of a nascent entrepreneur, Justin Kittredge, who used his experience and knowledge of the athletic footwear market to start his own footwear company, a manufacturer/distributor of customizable athletic sandals known as "slides." The case illustrates many of the concepts of Entrepreneurial Thought and Action-where an entrepreneur starts with the concepts of Who am I, What do I know, and Whom do I know.  |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
